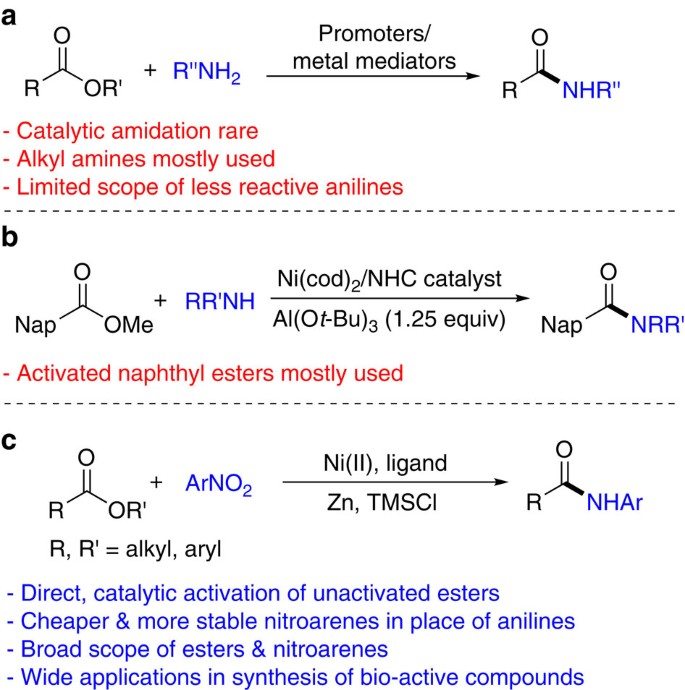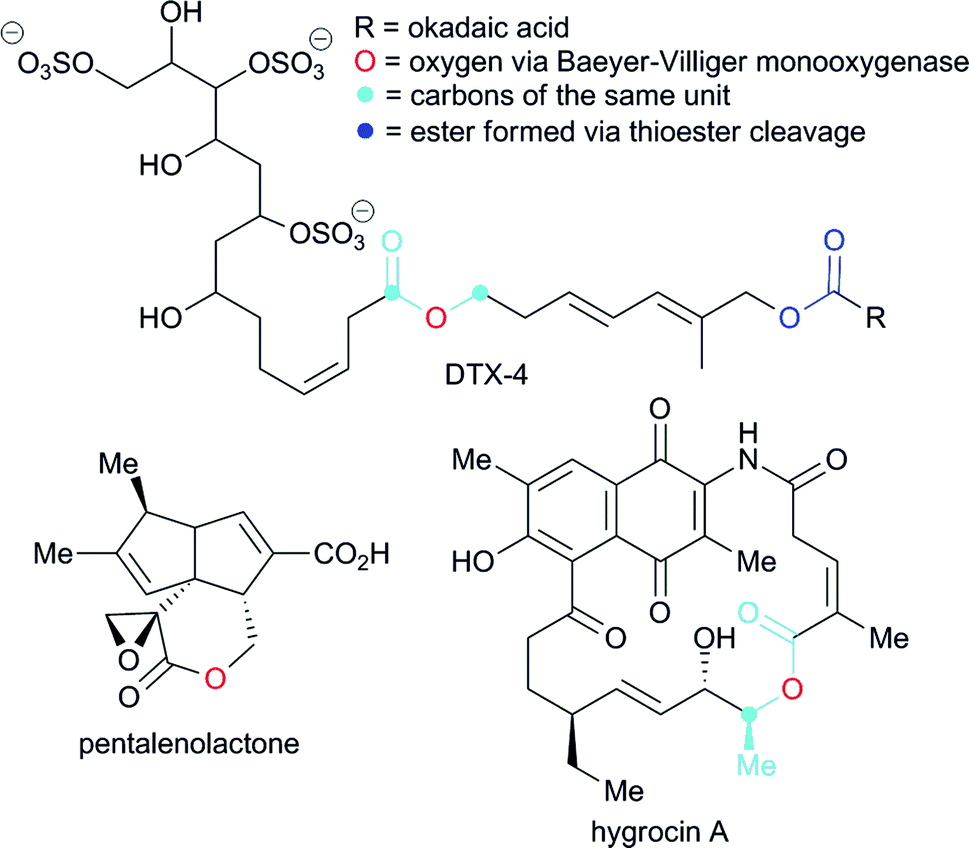
Ester coupling reactions – an enduring challenge in the chemical synthesis of bioactive natural products - Natural Product Reports (RSC Publishing)
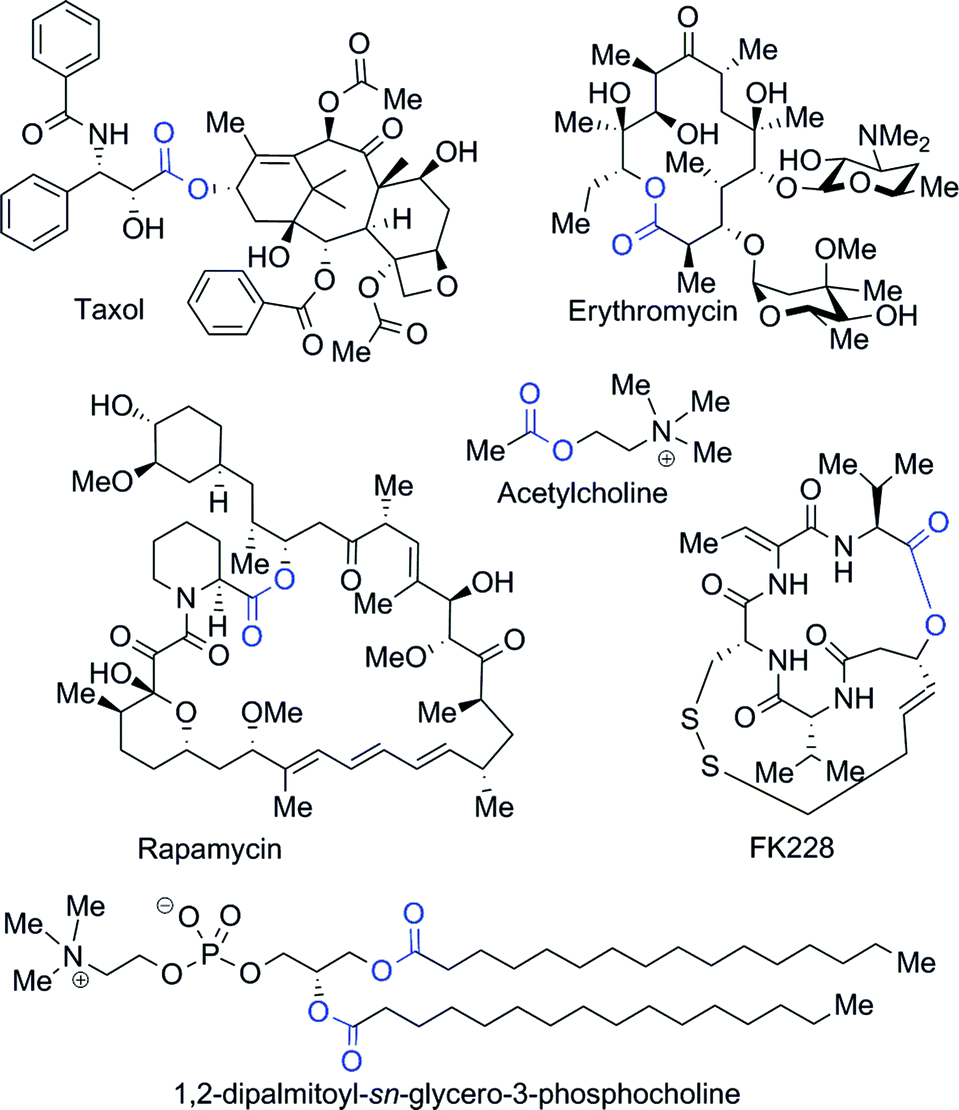
Ester coupling reactions – an enduring challenge in the chemical synthesis of bioactive natural products - Natural Product Reports (RSC Publishing)
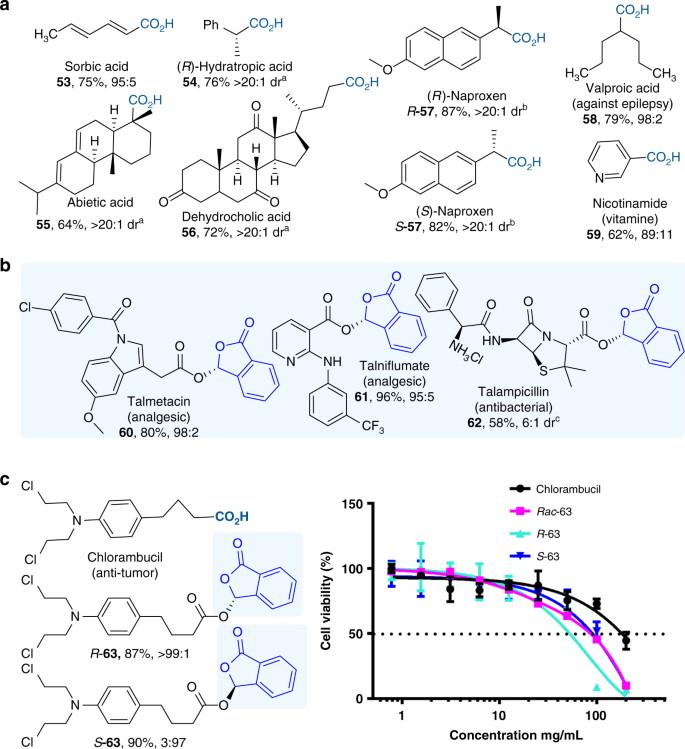
Catalytic asymmetric acetalization of carboxylic acids for access to chiral phthalidyl ester prodrugs | Nature Communications

Natural sucrose esters: Perspectives on the chemical and physiological use of an under investigated chemical class of compounds - ScienceDirect

Catalytic asymmetric acetalization of carboxylic acids for access to chiral phthalidyl ester prodrugs | Nature Communications
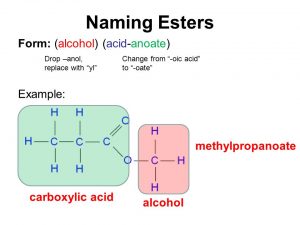
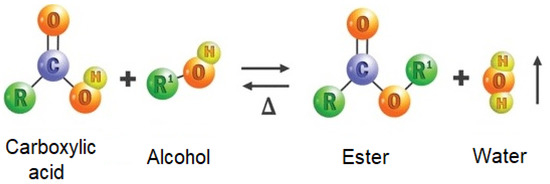







/ester-59134cd83df78c9283519859.png)